Standardized herbal extracts can interact with conventional medicines because they contain pharmacologically active compounds, but their consistent composition makes interaction risk easier to assess and control than with non‑standardized botanicals. When European buyers work with a quality‑focused supplier and robust documentation, herb–drug interaction risk becomes a manageable part of product design rather than an unknown.
This article is written for procurement, QA/RA, and formulation teams and does not provide medical advice for individual patients. Clinical decisions about medicines and herbal products must always sit with healthcare professionals.
What are standardized herbal extracts in B2B supply?
Standardized herbal extracts are plant‑derived preparations adjusted to contain defined levels of one or more marker compounds. These markers can be constituents with known biological activity or characteristic compounds used to control identity and strength. In B2B supply, standardization gives your teams a reproducible starting material with predictable potency from batch to batch.
In practice, a standardized extract is produced from controlled plant material under GACP and GMP‑aligned conditions, then tested with validated analytical methods. The specification will usually define a target assay range for key markers (for example, “24% flavone glycosides, 6% terpenes” for ginkgo) and set limits for impurities. Our standard herb extract portfolio reflects this approach, with each product supported by a Certificate of Analysis (CoA) and batch‑specific test results.
Marker compounds and consistency
Marker compounds sit at the core of any standardization strategy. They link the extract you buy today with the data generated in earlier studies, monographs, and internal evaluations. If the chemical profile stays within a defined range, you can apply existing knowledge about safety, interactions, and efficacy more confidently.
If you need a deeper dive on this concept, see our explainer on marker compounds in herbal extracts. For procurement and QA/RA teams, the key point is simple: without clear markers and robust assay methods, interaction data are much harder to interpret or apply to real products.
How standardization supports safer decisions
Standardization does not remove interaction risk, but it does reduce uncertainty. When active or marker constituents are controlled, observed herb–drug interactions from clinical or pharmacovigilance data become more relevant to your products. Your safety assessments, SmPC or label wording, and internal risk classifications can then build on a defined composition instead of a “black box” botanical.
This is especially relevant when you work within EMA/HMPC frameworks for herbal medicinal products. Consistent quality and composition make it easier to cross‑reference official monographs and EU dossiers when you prepare submissions or justify safety margins.
How do herb–drug interactions happen mechanistically?
Herb–drug interactions occur for the same reasons as drug–drug interactions: changes in exposure or overlapping pharmacological effects. For standardized extracts, these mechanisms remain relevant, even when quality is tightly controlled.
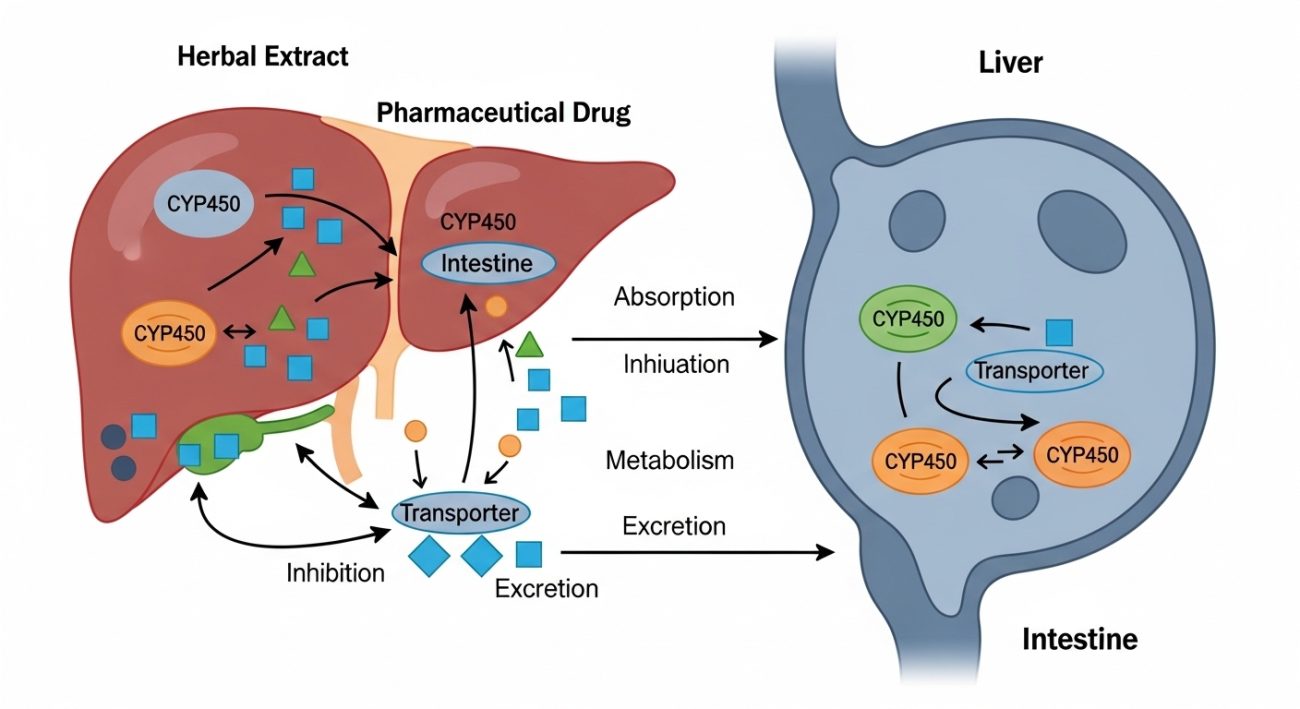
Pharmacokinetic interactions: enzymes and transporters
Pharmacokinetic (PK) interactions affect how the body absorbs, distributes, metabolizes, or eliminates drugs. Many small molecules are cleared by cytochrome P450 (CYP) enzymes and drug transporters such as P‑glycoprotein. Certain herbal constituents can inhibit or induce these systems.
Well‑known examples include extracts of St John’s wort, which can induce CYP3A4 and P‑glycoprotein, and green tea catechins, which can modulate transporters and enzymes in the gut. In practice, this may lower exposure to some drugs (reduced efficacy) or increase exposure (higher toxicity risk) when the drug has a narrow therapeutic index. PK interactions matter most in high‑dose standardized extracts used chronically alongside critical medicines.
Pharmacodynamic interactions: additive or opposing effects
Pharmacodynamic (PD) interactions arise when herbs and drugs act on similar physiological pathways. For example, botanicals with antiplatelet or anticoagulant activity may add to the effect of blood‑thinning medicines. Others may influence blood pressure, glucose regulation, or CNS function in the same direction as prescribed drugs.
Standardization can help here because it links PD effects more closely to dose and composition. However, your teams still need to consider additive or opposing mechanisms when designing indications, recommended intakes, and label warnings for finished products.
Real‑world variability and why it matters
Real‑world studies show wide variability in herbal products on the market. Unstandardized preparations may differ substantially in constituent levels between batches and brands. This variability makes interaction signals harder to interpret and can hide clinically relevant effects until they appear as spontaneous reports.
By contrast, standardized extracts reduce noise in these data. When exposure is constrained within a defined window, your pharmacovigilance and safety teams can more easily judge which interactions are plausible for your product and which are less likely.
Do standardized herbal extracts change interaction risk?
The short answer is yes and no. Standardization changes the profile of herb–drug interaction risk rather than eliminating it.
On the positive side, consistent marker levels mean that interactions identified in clinical trials or post‑marketing surveillance are more transferable. If your extract specification matches those used in published data, QA/RA teams can draw clearer conclusions. This applies across indications, from cardiovascular support to cognitive health.
On the other hand, standardization can also concentrate the constituents responsible for interactions. A high‑dose, standardized extract may produce stronger modulation of enzymes or transporters than a dilute traditional preparation. From a safety perspective, predictability improves, but the need for careful assessment remains.
Standardized versus non‑standardized extracts
When you compare standardized and non‑standardized extracts, three differences matter for interaction evaluation:
- Composition clarity: Standardized extracts have defined marker profiles; non‑standardized products may not.
- Batch‑to‑batch reproducibility: Tight assay ranges support reliable risk assessments.
- Regulatory alignment: Standardized profiles are easier to map to EMA monographs and dossier expectations.
For B2B buyers, this means standardized extracts offer a more robust platform for internal risk assessments, EU submissions, and communication with prescribers or pharmacists, even when the underlying interaction potential still exists.
Standardization does not remove interactions
No matter how well an extract is standardized, it still contains active molecules. If these molecules act on key enzymes, transporters, or receptors, interactions remain possible. This is why HMPC monographs and EU guidance documents often include specific warnings for certain herbal preparations, especially high‑dose forms of St John’s wort and other well‑studied botanicals.
A responsible supplier will therefore combine standardization with transparent communication about known interaction concerns, evidence gaps, and appropriate caution language for finished‑product labels.
Which standardized herbal extracts are high‑risk?
Only a subset of standardized herbal extracts show clinically important drug interactions. However, that subset includes several ingredients widely used in European nutraceutical and herbal medicinal products.
Examples often highlighted in scientific and regulatory discussions include:
- St John’s wort (Hypericum perforatum) – known for inducing CYP3A4 and P‑glycoprotein and reducing plasma levels of drugs such as certain immunosuppressants, anticoagulants, and antivirals.
- Ginkgo biloba – discussed in relation to antiplatelet and anticoagulant medicines.
- Echinacea purpurea – can modestly affect multiple CYP enzymes at high doses, with limited clinical impact but some concern around narrow‑therapeutic‑index drugs.
- Goldenseal (Hydrastis canadensis) – inhibits specific CYP isoenzymes in some studies.
These examples show why interaction risk should be evaluated per extract, not for “herbal products” as a single group.
Higher and lower concern categories
From a practical project viewpoint, you can group standardized extracts into:
- Higher‑concern group – robust literature on CYP or transporter modulation, or marked PD overlap with critical drug classes.
- Lower‑concern group – limited or mainly theoretical interaction signals, often based on high experimental doses or older studies.
Our dedicated articles on side effects of standardized herbal extracts and whether standardized herbal extracts are safe to use discuss broader safety aspects that sit alongside interaction considerations.
Evidence gaps and ongoing assessment
Many standardized extracts still lack large, controlled clinical studies focused specifically on herb–drug interactions. Evidence may come from in vitro experiments, small PK trials, or case reports. As EMA and national authorities stress, potential interactions should be assessed critically and balanced against the overall benefit–risk profile.
For B2B buyers, this means interaction risk assessment is an ongoing process. It should be updated as new data appear and as formulations, doses, and target populations evolve.
How should EU buyers evaluate interaction risk?
For European buyers working under EMA and HMPC frameworks, herb–drug interaction risk sits inside the broader quality and safety dossier. You need to align botanical ingredients with EU expectations while keeping commercial timelines realistic.
Mapping risk to EU regulatory expectations
EU guidance for herbal medicinal products emphasizes:
- High‑quality raw material under GACP
- Manufacturing under appropriate GMP for herbal APIs
- Clear definition of strength and composition
- Balanced assessment of potential interactions, especially where case reports or mechanistic data exist
When you assess a standardized extract, ask how easily it can be mapped to existing monographs, Ph. Eur. texts, or national guidance. Extracts that fit these structures usually integrate more smoothly into marketing authorization or notification pathways.
Supplier quality and testing that matter
Interaction risk assessment depends heavily on the underlying quality systems. Points to scrutinize include:
- Control of starting materials – traceable botanical source, contamination controls, and consistent harvesting practices.
- Extraction and standardization – validated processes to achieve defined marker ranges and remove unwanted components.
- Analytical testing – robust methods for assay, identity, contaminants, and, where relevant, interaction‑related parameters.
- Stability – data that show marker levels and impurity profiles remain within specifications through shelf‑life.
A supplier with established GACP/GMP alignment and transparent testing practices gives your QA/RA teams a stronger basis for interaction‑related decisions.
Documents to request from suppliers
To support interaction risk evaluation, procurement and QA/RA teams should request at least:
- Full CoA with marker assay, impurity profile, and microbial/heavy metal/solvent results
- Product specification and typical chromatographic fingerprints
- Technical data sheets summarizing known interaction concerns and evidence sources
- Stability summaries, especially for high‑dose or sensitive extracts
- For critical projects, any available in vitro CYP or transporter data
Our benefits of using standardized herb extracts article explains how these elements also support efficacy and branding, not only risk management.
Formulation considerations for interaction‑sensitive projects
Formulation choices can influence exposure to herbal constituents and, with it, interaction potential. This is particularly relevant when you use concentrated or novel delivery systems.
Dose, concentration, and exposure
High‑titer extracts deliver more active molecules per capsule or sachet than traditional herbal preparations. At the same time, modern formats often support chronic use. Together, this can increase the likelihood that interactions emerge, especially in populations on multiple medicines.
When your formulators work with standardized extracts, they should consider:
- Total daily intake of marker compounds
- Co‑administration with medicines that use the same metabolic pathways
- Intended treatment duration and expected user population
This does not mean such products are unsafe by default. It does mean the team should model realistic exposure scenarios and align these with known PK and PD mechanisms.
Liposomal and microencapsulated extracts
Liposomal and microencapsulated extracts may change the absorption profile of certain constituents. Higher or more consistent bioavailability can be desirable for efficacy, but it can also alter the balance of interaction risks.
In the absence of extensive clinical data, formulators should treat these systems with caution where co‑medication is likely. Early discussion with your supplier’s technical team can help you select appropriate extracts and doses for such formats.
Designing formulations around sensitive drugs
When your products target populations on narrow‑therapeutic‑index medicines, design choices matter even more. Options include:
- Selecting extracts with lower interaction concern based on current evidence
- Using conservative doses and clear on‑pack advice to consult healthcare professionals
- Documenting the rationale for ingredient selection and risk controls in your internal files
This design work is easier when the underlying extracts come from a partner who understands pharmacokinetic and regulatory implications and can adapt specifications to your needs.
Working with suppliers to manage interaction risks
Herb–drug interaction management is a shared responsibility between manufacturers, brand owners, and regulators. A capable standardized‑extract supplier should function as a technical partner, not only a bulk source.
Building an internal risk framework
Many European companies now treat herb–drug interactions under the same governance as other safety risks. A practical framework often includes:
- Initial screening of herbs and proposed indications for known interaction concerns
- Categorization of extracts into risk levels with corresponding documentation requirements
- Cross‑functional review between product development, QA/RA, pharmacovigilance, and medical affairs
- Periodic review as new evidence emerges or formulations change
Standardized extracts with clearly defined markers, strong documentation, and transparent testing slot more easily into this type of framework.
Key questions to ask a standardized extract supplier
During supplier qualification and project discussions, questions you might ask include:
- Which markers do you use for this extract, and how tight is the assay range?
- How do you control batch‑to‑batch variability that could affect interaction profiles?
- What do you know about this extract’s potential to affect CYP enzymes or transporters?
- How do you capture and communicate updates from the literature on safety and interactions?
- Can you support different grades or strengths for products aimed at more vulnerable user groups?
Answers to these questions help you judge whether the supplier can support your internal risk framework and EU documentation needs.
When to request samples, CoAs, and technical support
You should involve your supplier early whenever:
- You develop products for users on multiple medicines
- You plan to use high‑dose or novel formulations of botanicals with existing interaction concerns
- You aim for registration under EU herbal medicinal product pathways
At Xi’an B‑Thriving, our RFQ, sample, and COA process is designed to support this. Share your formulation brief, target market, and regulatory pathway, and our team can recommend suitable standardized extracts, provide documentation, and arrange samples for your internal evaluations.
Key takeaways for procurement, QA/RA, and formulators
For procurement, standardized extracts with tight specifications, proven quality systems, and transparent documentation reduce uncertainty around herb–drug interactions and help you qualify suppliers with confidence. For QA/RA teams, these extracts are easier to map to EMA/HMPC expectations and to integrate into dossiers, technical files, and pharmacovigilance plans.
For formulators, standardized extracts offer a more predictable building block. They still require careful design around sensitive drug classes and populations, but they allow clearer links between dose, exposure, and potential interactions. Across all functions, close collaboration with your extract supplier and early access to CoAs, specs, and technical data remain central to safe and successful product development.
If you are assessing standardized herbal extracts for a new EU project, consider aligning your ingredient choices with the safety principles discussed here and explore our standard herb extract range to identify candidates that match your quality and documentation needs.
FAQs on standardized herbal extracts and drug interactions
Do standardized herbal extracts interact with prescription medicines?
Yes, standardized herbal extracts can interact with prescription medicines because they contain active compounds that may affect enzymes, transporters, or pharmacological pathways. The benefit of standardization is that these potential interactions become more predictable and easier to evaluate in formal risk assessments.
Are standardized herbal extracts safer than traditional preparations for interactions?
Standardized extracts are not automatically “safer,” but they are easier to assess. Consistent marker levels and quality control let you link real‑world interaction data to the exact type of extract you use, while traditional preparations can vary more widely in composition.
Which standardized herbal extracts most often raise interaction concerns?
A small number of standardized herbal extracts frequently appear in interaction discussions, especially St John’s wort, ginkgo, and certain high‑dose preparations of other herbs. Most others show limited or mainly theoretical interaction potential, but each extract still needs individual evaluation.
How can formulators assess herb–drug interaction risks when choosing extracts?
Formulators can assess interaction risks by screening candidate herbs for known PK and PD mechanisms, reviewing monographs and regulatory guidance, and checking supplier documentation for composition and variability. Early collaboration with QA/RA and the supplier’s technical team helps translate this information into practical design choices.
What documentation should EU buyers request to evaluate interaction risks?
EU buyers should request full CoAs, specifications, chromatographic profiles, technical data sheets, stability information, and any interaction‑relevant in vitro or clinical summaries the supplier can share. These documents support both internal risk frameworks and EMA‑aligned dossiers.
Does standardization of marker compounds eliminate herb–drug interactions?
No, standardization does not eliminate herb–drug interactions. It reduces uncertainty by controlling composition, but the underlying active molecules still have the potential to interact with drugs, especially at higher doses or in sensitive user groups.
Do liposomal or microencapsulated herbal extracts increase interaction risk?
Liposomal and microencapsulated extracts may change absorption and exposure profiles, which can influence interaction potential. Without extensive clinical data, they should be used with careful dose selection and clear risk assessments, particularly for products aimed at users on long‑term medication.
Are standardized herbal extracts safe to use in general?
Standardized herbal extracts are designed to offer consistent quality and predictable performance, which supports safer use in regulated products. However, overall safety, including interactions, depends on the specific extract, dose, user population, and co‑medication, all of which must be evaluated by your internal experts; our detailed safety guide for standardized herbal extracts covers this in more depth.

