
Standardized to 95% curcuminoids means that a turmeric extract has been scientifically processed to ensure a consistent concentration of active compounds. While raw turmeric root typically contains only 3% to 5% curcuminoids, a 95% standardized extract is a highly concentrated “monomer” or “enriched extract.” This specification guarantees that every kilogram of bulk powder contains exactly 950 grams of the three primary active molecules: curcumin, demethoxycurcumin, and bisdemethoxycurcumin.
For procurement managers and formulators, this standardization is a benchmark for quality and dosage precision. It ensures that the final nutraceutical or pharmaceutical product delivers a predictable physiological effect, regardless of the harvest conditions of the raw turmeric.
The Science Behind 95% Curcuminoid Standardization
Standardization is the process of bringing a botanical ingredient to a specific chemical marker level. In the context of Curcuma longa, the focus is on the group of polyphenols known as curcuminoids. Achieving a 95% threshold requires sophisticated solvent extraction and crystallization techniques. This process removes the starches, fibers, and essential oils found in the raw root, leaving behind a refined crystalline powder.
When you evaluate a standardized herbal extract, you are looking for chemical uniformity. This uniformity allows product development leads to calculate precise dosages for capsules, tablets, or functional foods. Without this 95% guarantee, a formulator would struggle with significant “assay drift,” where one batch of product is significantly weaker than the next.
Understanding the Three Core Curcuminoids
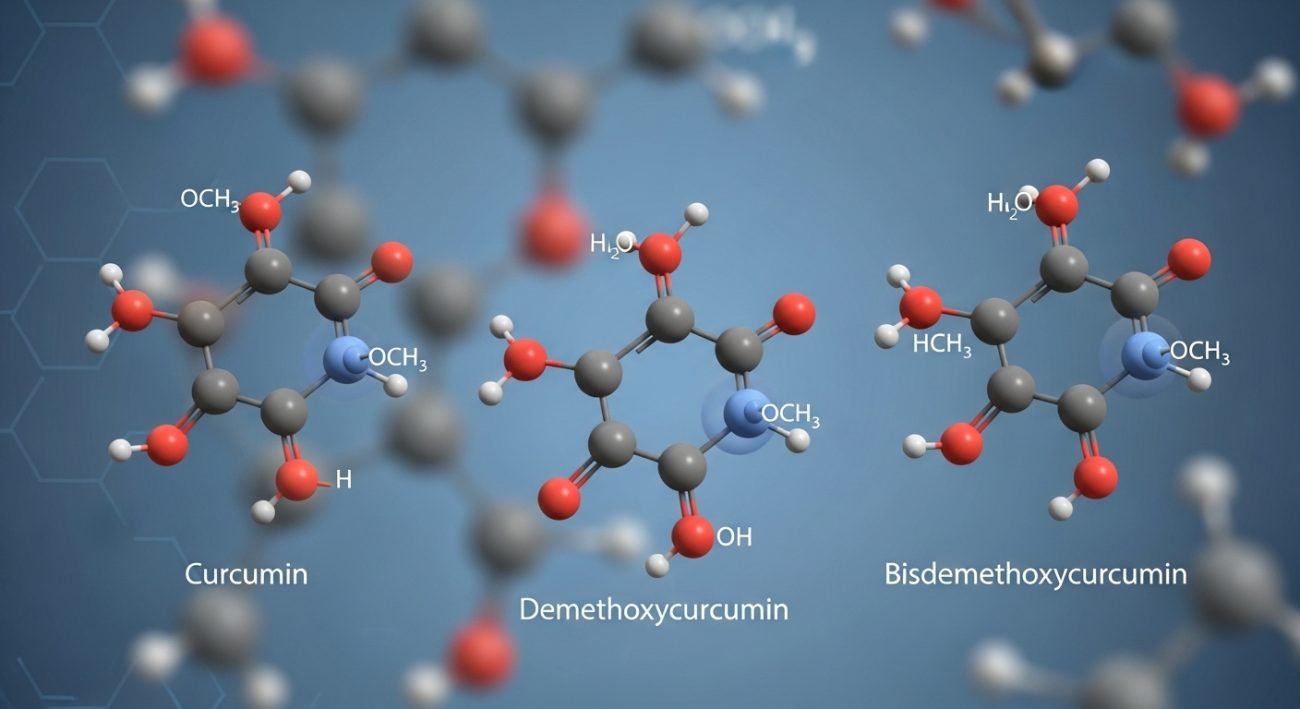
A common misconception in B2B procurement is that “95% Curcumin” refers to a single molecule. In reality, “95% Curcuminoids” refers to a specific complex of three distinct compounds. A high-quality Certificate of Analysis (COA) should ideally break down these three components:
- Curcumin (Curcumin I): Usually the most abundant, typically making up 70–80% of the complex.
- Demethoxycurcumin (Curcumin II): Typically represents 15–20%.
- Bisdemethoxycurcumin (Curcumin III): Usually found in the 2–5% range.
The synergy between these three molecules is what European regulatory bodies and pharmacopoeias (such as the USP or EP) recognize as the authentic profile of a turmeric extract.
HPLC vs. UV Testing Methods for Purity
The method used to verify the 95% concentration is just as important as the number itself. Historically, many suppliers used UV-Vis (Ultraviolet-visible spectroscopy) to test curcuminoids. However, UV testing can be easily “fooled” by synthetic dyes or other polyphenols, often leading to inflated results.
At Xi’an B-Thriving, we prioritize High-Performance Liquid Chromatography (HPLC). HPLC is the gold standard for European QA/RA managers because it separates each curcuminoid. This method provides a “fingerprint” of the ingredient, ensuring the 95% claim is accurate and free from adulteration. When requesting a sample, always ask for the HPLC chromatogram to confirm the purity of the batch.
Why 95% is the Industry Standard for B2B Procurement
The transition from raw turmeric powder to a 95% extract is a matter of efficacy and formulation space. Raw turmeric is excellent for culinary use, but it is inefficient for therapeutic applications. To achieve the curcuminoid levels found in a single 500mg capsule of 95% extract, a consumer would need to ingest nearly 15 grams of raw turmeric powder.
For manufacturers, the herbal extract standardization process offers three primary advantages:
- Dose Consistency: You can guarantee the active content on your supplement facts panel.
- Stability: Concentrated curcuminoids are more stable over time than raw plant material.
- Cost-Efficiency: Although the price per kilo is higher for extracts, the “price per active milligram” is significantly lower due to the extreme concentration.
How to Verify Quality in a Curcumin 95% COA
A B2B buyer should look beyond the “95%” figure on a specification sheet. European compliance standards, particularly for the nutraceutical and pharma sectors, require strict adherence to safety limits. A comprehensive COA from a reliable manufacturer must include several critical parameters.
First, check the Solvent Residue. Since 95% curcuminoids are produced via solvent extraction (often using ethanol or ethyl acetate), the levels must remain well below the limits defined by EU Directive 2009/32/EC. Second, verify the Heavy Metal Profile. Turmeric is a bio-accumulator; therefore, Lead, Cadmium, Mercury, and Arsenic levels must be strictly monitored to meet European safety standards. Finally, ensure the Microbial Load is within the limits for “Ready-to-Use” dietary ingredients to avoid secondary contamination during production.
Formulating with Standardized 95% Curcuminoid Powder
Integrating 95% curcuminoids into a product line requires an understanding of its physical properties. The powder is intensely hydrophobic (water-insoluble) and possesses a vibrant orange-yellow pigment. These characteristics influence how you approach your delivery system.
For traditional capsules and tablets, 95% powder is the industry baseline. However, if you are developing a functional beverage or a high-absorption supplement, you may need to consider secondary processing. Xi’an B-Thriving supports these advanced needs through liposomal or microcapsule technologies, which enhance the solubility and bioavailability of the 95% standardized material.
Frequently Asked Questions (FAQ)
Is 95% curcumin the same as 95% curcuminoids?
Technically, no, though the terms are often used interchangeably in trade. “Curcuminoids” refers to the group of three active molecules, while “Curcumin” is just one of those three; a 95% standardized extract should correctly be labeled as “Total Curcuminoids.”
What solvents are used to reach a 95% concentration?
Food-grade ethanol or ethyl acetate are the most common solvents used in high-purity extractions. Reputable manufacturers perform a final purification step to ensure any residual solvents are well within EU safety limits.
Does 95% standardization guarantee high bioavailability?
Standardization guarantees potency and purity, but not necessarily absorption. Because curcuminoids are fat-soluble, many formulators combine 95% powder with lipids or piperine (black pepper extract) to improve metabolic uptake.
How should 95% curcuminoid powder be stored in bulk?
The powder is sensitive to light and high temperatures. To maintain the 95% assay over its shelf life, it should be stored in a cool, dark place in well-sealed, UV-resistant packaging, typically in fiber drums with double plastic liners.
Can I get a sample and a COA before a bulk order?
Yes, providing a batch-specific Certificate of Analysis and a pre-shipment sample is standard practice for B2B botanical manufacturing. This allows your QA team to verify the 95% HPLC assay in-house before committing to a large-scale procurement.
Are you looking to secure a consistent supply of high-purity turmeric extract?
Xi’an B-Thriving provides 95% Curcuminoid powder with full HPLC documentation and EU-compliant testing. Contact our technical team today to request a formal RFQ, a digital COA, or a 50g sample for your next formulation trial.

