A herb extract is standardized by calibrating the manufacturing process so that every batch delivers a defined, measurable concentration of a specific marker compound. The process spans botanical sourcing, extraction, purification, analytical testing, and batch verification — with each step documented and confirmed in a Certificate of Analysis (COA).
For European procurement managers and formulators sourcing at scale, understanding this process is the foundation of qualified supplier selection. A supplier who can explain and document every stage is one worth trusting.
What “Standardization” Really Means in Herbal Extraction
Standardization is a quality assurance discipline, not a single test. It means a manufacturer has identified one or more marker compounds in a botanical raw material and set a reproducible specification — typically expressed as a minimum percentage — that every production batch must meet.
The word “standardized” does not automatically mean the extract is more potent or more bioavailable. It means the concentration of a chosen marker is consistent and verifiable. That distinction matters when you source ingredients for EU-regulated nutraceutical, pharma, or cosmetic formulations.
Standardized Extract vs. Ratio Extract: What Buyers Must Know
These two terms describe fundamentally different things, and confusing them carries real procurement risk.
A ratio extract (for example, 10:1) states that ten kilograms of dry herb produced one kilogram of extract. It says nothing about the concentration of any active compound. Two 10:1 extracts from different suppliers may carry completely different phytochemical profiles, depending on raw material quality and processing variables.
A standardized extract specifies the measured concentration of a defined marker compound — for example, 95% curcuminoids or 24% flavonoid glycosides. When a COA confirms this specification, you have a quantified basis for formulation decisions. For EU buyers building compliant product dossiers, this distinction is non-negotiable.
Active Markers, Analytical Markers, and Fingerprint Standards Explained
The European Medicines Agency (EMA) distinguishes three types of standardization markers. Understanding which type your supplier reports affects both your label claims and regulatory documentation.
- Active markers — compounds with recognized pharmacological relevance to the botanical’s application, such as withanolides in ashwagandha
- Analytical markers — compounds used for identification and quantification purposes, which may not themselves be the primary bioactive agents (hypericin in St. John’s Wort is a common example)
- Fingerprint standards — chromatographic profiles confirming extract identity and batch consistency without isolating a single compound
The phrasing on your COA — “assayed by HPLC as…” — indicates which type applies. EU QA/RA teams should clarify this before supplier approval.
The Standardization Process: From Raw Herb to Certified Extract
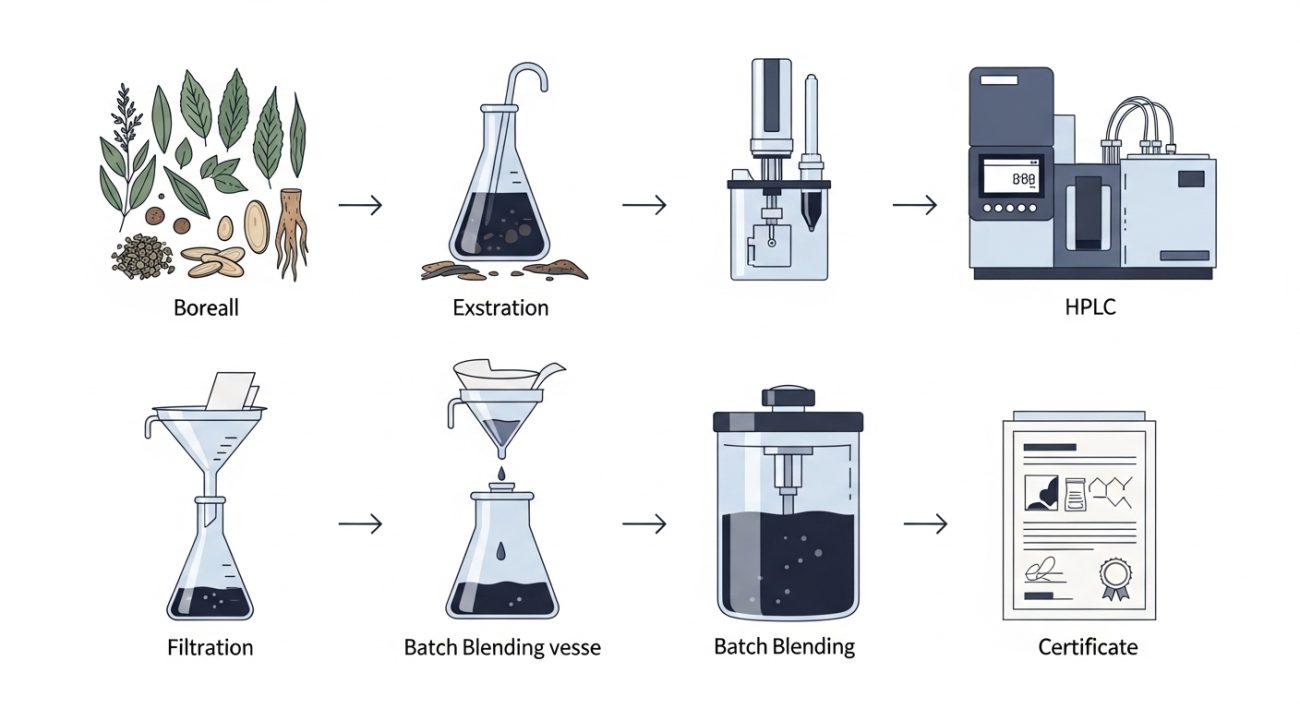
Standardization runs through the entire manufacturing chain, not just the final QC step. Here is how a rigorous supplier approaches it, from the field to the finished batch.
Step 1: Botanical Sourcing and Species Authentication
Correct botanical identity must be confirmed before extraction begins. Suppliers use macroscopic and microscopic analysis, thin-layer chromatography, or DNA barcoding depending on the herb. Adulterants and species substitutions remain a documented quality risk in bulk botanical supply chains.
Manufacturers sourcing from contracted cultivation programs or vertically integrated supply chains maintain better control over agrochemical residue levels and harvest-time marker concentrations — both factors that directly affect final extract quality.
Step 2: Selecting the Right Extraction Solvent and Method
Solvent choice controls which compounds extract and in what proportion. Ethanol-water blends extract a broad range of polar and semi-polar compounds and remain the most widely used option. Supercritical CO₂ extraction suits lipophilic actives such as β-carotene. Water extraction targets polysaccharide-rich material.
For EU supply, solvent selection has a regulatory dimension. ICH Q3C classifies residual solvents by toxicological risk. Class 3 solvents such as ethanol carry the lowest concern. If a Class 2 solvent such as methanol was used at any production stage, the COA must include residue testing results confirming it falls within permitted limits.
Step 3: Concentration, Purification, and Enrichment
After initial extraction, the liquid undergoes concentration — typically via vacuum evaporation — to remove solvent and increase marker density. Many suppliers then run selective purification steps such as resin adsorption or column chromatography to eliminate chlorophyll, heavy metals, or unwanted co-extracted compounds.
This stage is where high-specification extracts are produced: 95% curcuminoids, 80% silymarin, and similar. Without this step, the result is a broad-spectrum extract with variable and typically low marker concentrations.
Step 4: Analytical Testing to Confirm Marker Compound Levels

Once the dried extract is ready, analytical testing confirms whether the batch meets specification. High-performance liquid chromatography (HPLC) is the industry standard. It separates compounds through a stationary phase interaction and converts the area-under-curve data from each peak into percentage concentrations.
Suppliers should run HPLC both in-house — for process control — and through an accredited third-party laboratory for COA credibility. If a supplier cannot provide a third-party HPLC report on request, treat that as a qualification risk.
Step 5: Batch Homogenization for Consistency Across Lots
Natural raw materials vary by season, geography, and growing conditions. Even with tight extraction controls, individual production lots can show marker concentration variation. Responsible manufacturers blend compatible lots to produce a homogeneous final batch that meets specification throughout — not just in a spot sample.
This step rarely appears in supplier marketing content, but formulators who need lot-to-lot consistency across production runs should ask about it directly.
Step 6: Certificate of Analysis Issuance and What It Contains
The COA is the formal document confirming every batch meets its full specification. A complete COA for a standardized herbal extract should include:
- Botanical identity — species name, plant part, extraction solvent
- Active/analytical marker assay result — percentage, confirmed by HPLC or UV-Vis
- Heavy metal limits — lead, arsenic, mercury, cadmium against EP or USP thresholds
- Microbial count results — total aerobic count, yeast/mould, and specified pathogen absence
- Solvent residue analysis — with ICH Q3C class reference
- Batch number and manufacture/expiry dates
- Testing methodology references
For a detailed walkthrough of what to look for in supplier documentation, see our guide on how to verify a COA for herbal extracts.
Analytical Methods Used in Herbal Extract Standardization
HPLC and HPTLC: Quantifying Active Markers with Precision
HPLC remains the primary quantitative method across the industry. It separates compounds by polarity interaction and reports concentrations with high reproducibility. Most European Pharmacopoeia monographs specify HPLC as the reference method for marker assay.
HPTLC (High-Performance Thin-Layer Chromatography) serves identity confirmation and fingerprint analysis more commonly than precise quantification. Some suppliers use it as a rapid screen before full HPLC. Both methods generate chromatographic data that suppliers can share with buyers as supporting documentation alongside the COA.
Microbial, Heavy Metal, and Solvent Residue Testing for EU Markets
EU buyers face stricter import documentation requirements than most other markets. European Pharmacopoeia standards, combined with food supplement and cosmetic ingredient regulations, require documented evidence on three fronts:
- Heavy metals — typically Lead ≤3 ppm, Arsenic ≤1 ppm, Mercury ≤0.1 ppm (thresholds vary by application and applicable regulation)
- Microbial contamination — total aerobic count, confirmed absence of E. coli, Salmonella, and Staphylococcus aureus
- Solvent residues — Class 2 solvents require ICH Q3C limit confirmation; Class 3 solvents need confirmation that testing was performed
A supplier who cannot provide these results in a structured COA creates downstream compliance risk for the importing company.
EU Regulatory Standards for Standardized Herbal Extracts
European Pharmacopoeia and EMA Specifications: Core Requirements
The European Pharmacopoeia (EP) publishes monographs covering many commonly traded herbal substances and preparations. Monograph 0765 (Herbal Drug Preparations) establishes general requirements covering definition, production, identification, and testing for herbal extracts entering EU markets.
The EMA’s guideline on specifications for herbal substances distinguishes between active and analytical markers and sets principles for acceptable batch specifications. Suppliers exporting to EU-regulated manufacturers should demonstrate familiarity with these frameworks — even where their primary compliance is under Chinese national standards such as GB or GACP.
What EU Buyers Should Request from Any Standardized Extract Supplier
Before approving a new supplier, EU procurement teams should request the following:
- Full COA with third-party analytical backing
- Applicable certifications: GMP (ISO 22000, FSSC 22000, or pharmaceutical GMP equivalent), plus Halal or Kosher certificates where product scope requires them
- Residual solvent report with ICH Q3C class references
- Heavy metal report aligned to EP or USP limits
- Stability or shelf-life testing data
- A physical sample with matching COA for independent laboratory verification
Independent lab testing of the sample against the supplier’s COA remains the most reliable qualification step available before committing to bulk volume.
Standardization Benchmarks for Commonly Sourced Botanicals
Industry-Accepted Marker Levels for High-Demand Extracts
These are the standardization levels most commonly referenced in European B2B formulation and procurement. Use them as reference points when reviewing a supplier’s offered specification.
| Botanical | Marker Compound | Common Standard |
|---|---|---|
| Turmeric (Curcuma longa) | Curcuminoids | 95% |
| Milk Thistle (Silybum marianum) | Silymarin | 70–80% |
| Ashwagandha (Withania somnifera) | Withanolides | 2.5–5% |
| Ginkgo Biloba | Flavonoid glycosides / Terpene lactones | 24% / 6% |
| Green Tea (Camellia sinensis) | EGCG / Total polyphenols | 45–98% |
| Grape Seed (Vitis vinifera) | Oligomeric proanthocyanidins (OPCs) | 95% |
| Panax Ginseng | Ginsenosides | 5–80% |
These figures represent industry-typical benchmarks. Always confirm the actual specification against the supplier’s COA before finalizing a formulation.
Why Standardization Levels Can Vary Between Manufacturers
Two suppliers can both label a product “5% withanolides” and deliver extracts with meaningfully different analytical profiles. The reasons include differences in raw material cultivar, harvest timing, extraction solvent ratios, purification depth, and the HPLC reference standard used for calibration.
This is the phytoequivalence challenge: a standardized extract is reproducible within one supplier’s own system but is not automatically equivalent to another supplier’s extract carrying the same nominal specification. Formulators relying on published research tied to a specific extract should verify both the production method and the analytical reference standard in use.
How B-Thriving Standardizes and Certifies Every Bulk Batch
Our QC Workflow: In-House Testing and Third-Party Verification

At Xi’an B-Thriving, every standardized extract batch goes through a dual-verification workflow. In-house HPLC and UV-Vis spectrometry testing runs throughout production for real-time process control. Before any batch ships, an accredited third-party laboratory confirms the final marker assay and generates the COA that accompanies every order.
We provide full documentation packages to EU buyers: COA, heavy metal report, microbial analysis, solvent residue results, and applicable certification copies. Our technical team is available to discuss specification alignment, EU documentation requirements, and formulation compatibility before you place an order.
Browse confirmed-specification botanicals in our standardized herbal extract catalog.
Request a COA, Sample, or Bulk RFQ for Your Formulation
If you are evaluating a new botanical ingredient or comparing supplier quality, the most direct path forward is to request a sample and COA together. We supply pre-shipment samples with full analytical documentation, and we can prepare custom specification quotes for non-standard standardization levels or OEM requirements.
To request a COA, product sample, or bulk RFQ — provide your target herb, marker specification, and required volume — and our team will respond within one business day.
Frequently Asked Questions
What is the difference between a standardized extract and a 10:1 ratio extract?
A standardized extract guarantees a measured concentration of a specific marker compound confirmed by analytical testing. A 10:1 ratio extract only indicates that ten parts of raw herb produced one part of extract — it confirms nothing about active compound concentration.
Which analytical method do suppliers use most to verify herbal extract standardization?
HPLC is the industry standard. It quantifies marker compounds with high precision and is referenced in most European Pharmacopoeia monographs as the preferred assay method for herbal extracts.
What should a COA for a standardized herbal extract always include?
A complete COA covers botanical identity, active marker assay result with method stated, heavy metal limits, microbial counts, solvent residue analysis, batch number, and manufacture/expiry dates.
Why do standardization levels for the same herb vary between manufacturers?
Differences in raw material cultivar, extraction solvent, purification depth, and the HPLC reference standard used for calibration all affect the final marker percentage. Two extracts carrying identical label specifications may not be analytically equivalent.
What EU regulations apply to standardized herbal extract imports?
The European Pharmacopoeia — particularly Monograph 0765 — and EMA guidelines on herbal substance specifications set the quality framework. EU Directive 2001/83/EC applies when extracts enter medicinal product manufacturing. ICH Q3C governs solvent residue limits for human-use applications.
How do I qualify a new herbal extract supplier for EU supply?
Request a COA backed by third-party analytical testing, confirm available GMP certifications, review heavy metal and solvent residue reports against EP/USP limits, and obtain a sample for independent laboratory verification before placing bulk orders.

