Turmeric standardized extract benefits are easiest to understand when you connect three things: chemistry, clinical evidence, and quality control. A standardized extract gives you a repeatable level of curcuminoids (main actives), which helps brands design consistent formulas and reliable labeling. It also simplifies procurement, COA review, and batch-to-batch performance.
What is a turmeric standardized extract, and why does standardization matter?
A turmeric standardized extract is a turmeric (Curcuma longa) extract that is tested and adjusted to hit a defined marker level, usually total curcuminoids. Standardization matters because turmeric varies by origin, harvest, and processing. A fixed spec supports consistent dosing, predictable color/odor, and easier regulatory and QA workflows.
What does “95% curcuminoids” actually mean in practice?
“95% curcuminoids” usually means the extract contains about 95% of the three main curcuminoids (curcumin, demethoxycurcumin, bisdemethoxycurcumin), measured by an analytical method such as HPLC. It does not mean 95% whole turmeric. It’s a concentrated fraction designed for potency and repeatability.
Quick clarity (buyers love this table):
| Label phrase | What it typically refers to | What to confirm on COA |
|---|---|---|
| “Total curcuminoids X%” | Sum of 3 curcuminoids | Method (HPLC), reference standard, calculation basis |
| “Curcumin X%” | Curcumin only | Whether DMC + BDMC are also reported |
| “Turmeric extract 10:1” | Drug-extract ratio (DER) | DER definition + solvent + marker assay |
| “Full spectrum turmeric” | Broader compound profile | Marker level + fingerprint + adulteration checks |
If you want a deeper definition of standardization for buyers, see Standard Herb Extract.
Which tests should appear on a turmeric standardized extract COA?
A strong COA should show identity, potency, safety, and contamination controls. At minimum, you want curcuminoid assay, microbiology, heavy metals, residual solvents (if used), and adulteration screening where relevant. This is the fastest way to reduce complaints and returns at scale.
COA checklist (copy/paste for procurement):
- Identity: botanical ID + chromatographic fingerprint
- Assay: total curcuminoids (and ideally each curcuminoid)
- Heavy metals: Pb, Cd, As, Hg (limits aligned with your market)
- Micro: TAMC/TYMC, pathogens (e.g., E. coli, Salmonella)
- Residual solvents: aligned to ICH Q3C if solvent extraction is used
- Adulteration risk controls: screening for lead chromate where risk is elevated
What benefits does turmeric standardized extract offer based on human evidence?
Human studies suggest turmeric extracts and curcuminoids can support inflammation balance, joint comfort, cardiometabolic markers, and liver fat markers in certain groups. Results vary by dose, formulation, and study quality. The most consistent signals show up in joint osteoarthritis outcomes and inflammatory biomarkers, but claims still need careful wording.
Which benefits have the strongest clinical signal today?
Joint discomfort in knee osteoarthritis has the clearest body of evidence among common use-cases. Multiple meta-analyses report improvements in pain and function versus placebo. Many analyses still rate certainty as low-to-moderate because trials differ in design and dosing. Still, it’s the most repeatable benefit area for product strategy.
Evidence snapshot (brand planning view):
| Benefit area | What trials commonly measure | Typical signal | Notes for claims |
|---|---|---|---|
| Joint comfort (OA) | WOMAC, VAS pain | Often improves | Avoid disease treatment claims |
| Inflammation markers | CRP, TNF-α, IL-6 | Often decreases | Useful for “supports healthy inflammation response” |
| Lipids | TC, LDL-C, TG | Modest improvements | Often stronger in metabolic conditions |
| NAFLD markers | ALT/AST, steatosis indices | Mixed to positive | Subgroup effects may appear |
| Mood symptoms | depression/anxiety scales | Emerging | Needs careful positioning |
How does turmeric standardized extract support joint comfort?
Many RCTs in knee osteoarthritis use curcumin/curcuminoids and report lower pain scores and better function scores compared with placebo. Meta-analyses commonly conclude curcuminoids can help as an adjunct option, though study quality varies. Position it as support for mobility and comfort, not as a cure for arthritis.
Practical product angle (what brands usually do):
- Choose a standardized curcuminoids spec (often 95% or a lower standardized extract).
- Consider a bioavailability approach if your dose target is modest.
- Build claims around comfort, mobility, and activity support.
What does the evidence say about inflammation and oxidative stress markers?
Across intervention studies, curcumin supplementation often reduces biomarkers like CRP, TNF-α, and IL-6, and may influence oxidative stress markers. Meta-analytic findings generally support an anti-inflammatory direction, but effect sizes vary by population and formulation. For marketers, this supports “healthy inflammatory response” language with appropriate disclaimers.
Can turmeric standardized extract support heart and metabolic health markers?
Umbrella reviews and meta-analyses report modest improvements in lipid profiles in some populations, especially those with metabolic conditions. Effects are not uniform, and study designs differ. Standardized extracts help because they provide a consistent dose and clearer QC for long-term product quality.
What benefits are seen for fatty liver (NAFLD) markers?
Systematic reviews in NAFLD populations suggest curcumin can improve some liver-related outcomes in certain subgroups, such as liver enzymes or steatosis-related measures. Results can be inconsistent across trials, which makes formulation choice and study-matching important. This is a promising but careful category for brands.
Does turmeric standardized extract help mood and stress outcomes?
Meta-analyses in mood-related outcomes suggest potential improvements in depressive symptoms and anxiety measures in some studies. The evidence is growing, but it still varies across populations and curcumin formats. Brands typically position this as “supports emotional wellbeing” and avoid medical claims unless operating under a drug framework.
Why is curcumin hard to absorb, and what does that mean for benefits?
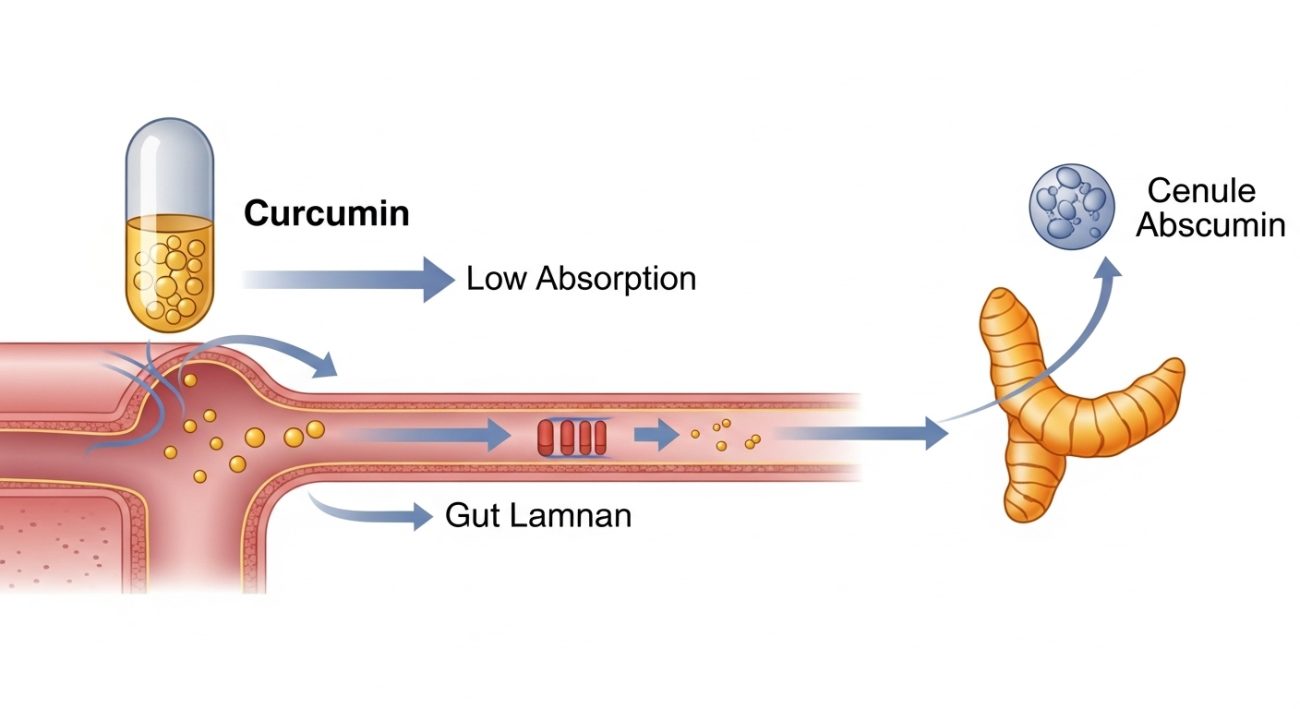
Curcumin has low oral bioavailability due to poor solubility and rapid metabolism. That means a high label dose does not always translate into high systemic exposure. Standardization ensures consistent potency, but absorption still depends on formulation. This is why delivery systems and absorption enhancers are common in turmeric products.
What role does black pepper (piperine) play in absorption?
Piperine can significantly increase measured curcumin bioavailability in human pharmacokinetic research. That benefit also raises a real-world concern: higher exposure can increase interaction risk for some consumers. Brands often use piperine for efficacy-per-dose, but they should also tighten safety language and QA documentation.
Tip: If you use piperine, build a stronger “Who should avoid this?” FAQ for your product page, especially for medication users.
What “bioavailability-enhanced” turmeric formats exist?
Common approaches include phospholipid complexes, micelles, nanoparticles, and liposomal systems. These aim to increase absorption and circulating levels. They can also change safety profiles, which is why highly bioavailable curcumin products may need stronger safety review than culinary turmeric.
How do you choose the right turmeric standardized extract for your product?
Choose based on your target benefit, dose strategy, dosage form, and regulatory environment. A standardized extract simplifies consistent dosing and supports stable sensory performance. The best choice is often the one that matches the strongest evidence category for your claim, not simply the highest percentage.
Which specification questions should procurement ask before buying?
Procurement should ask questions that force clarity on potency, safety, and traceability. This reduces reformulation cycles and protects your brand during audits. Treat turmeric like a “high-risk colorant botanical” from a QA perspective, not a simple spice.
Procurement spec template:
| Spec category | Ask for | Why it matters |
|---|---|---|
| Potency | Total curcuminoids + individual breakdown | Prevents spec gaming |
| Method | HPLC method + reference standards | Confirms comparability |
| Safety | Heavy metals + micro + residual solvents | Market compliance |
| Origin | Country + processor traceability | Risk management |
| Adulteration | Lead chromate risk controls | Protects consumers |
How do brands match extract types to applications?
Standardized extracts behave differently in capsules, tablets, gummies, and beverages. High-curcuminoid extracts can be intense in color and taste. Water-dispersible systems can help beverages, but they may cost more. Capsules remain the simplest delivery format for potency-driven positioning.
Practical matching:
- Capsules/tablets: high-curcuminoid standardized extracts
- Gummies: taste masking + stability becomes the main constraint
- Beverages: water-dispersible or emulsified curcumin systems
To help readers understand the difference between broader botanical profiles and marker-driven consistency, see Full Spectrum Extract vs Standardized Extract.
What safety, interaction, and regulatory realities shape turmeric standardized extract benefits?
Turmeric is widely used, but supplements are not risk-free. Evidence can support product rationale, while regulations control claims. Always align your claims with your market’s rules, and treat safety and contamination control as core quality requirements.
What does European traditional-use guidance say about turmeric preparations?
European herbal monographs recognize turmeric preparations for traditional use in mild digestive disturbances, such as fullness and flatulence. They also warn about bile-duct obstruction and gallbladder-related conditions due to effects on bile secretion. This matters for label cautions and customer-facing safety FAQs.
Can brands claim “joint function” for curcumin in the EU?
EFSA has evaluated specific health claims such as “normal functioning of joints” for curcumin and concluded a cause-and-effect relationship was not established for that claim in the evaluated context. Brands selling in the EU should avoid prohibited health-claim wording in foods and supplements.
What quality risks exist in turmeric supply chains?
Turmeric faces a known risk of economically motivated adulteration, including lead chromate to intensify yellow color. Brands mitigate this with vendor qualification, heavy metal testing, and targeted adulteration screening. Treat strong heavy-metal controls as essential, not optional.
Why are liver-related safety discussions increasing around curcumin supplements?
Safety reviews note case reports and clusters of liver injury linked to some turmeric/curcumin supplements, especially when absorption enhancers are involved. This does not mean turmeric is inherently unsafe, but it does mean high-bioavailability formats should get extra caution, clearer warnings, and stronger post-market monitoring.
How can formulators make benefits more consistent in real products?
Consistency comes from controlling potency, dispersion, stability, and dose delivery. Standardization gives you the same active per gram. Formulation gives you the same exposure per serving. If either breaks, customer experience becomes inconsistent.
Formulation tips that usually save time:
- Test for staining and color drift early in development.
- Use fat-compatible systems for better dispersion in some formats.
- Plan for taste masking if you use high-potency extracts.
- Validate assay after processing, not only at raw material stage.
Which FAQs help you win Featured Snippets and reduce buyer friction?
Is turmeric extract the same as curcumin?
Not exactly. Turmeric extract can contain many compounds from turmeric, while curcumin refers to one major curcuminoid. Many standardized extracts focus on total curcuminoids, which include curcumin plus related curcuminoids. Always confirm what your spec measures and what your label claims.
What is a good standardized percentage for most supplement products?
There is no single best percentage. Many products use high-curcuminoid extracts for potency, while others use lower standardized extracts to balance taste, cost, and formulation constraints. The best percentage is the one that matches your target evidence area, serving size, and claim strategy.
Should brands prefer standardized extracts or full-spectrum turmeric?
Standardized extracts offer repeatable dosing and simpler QA. Full-spectrum options may offer a broader phytochemical profile but can vary more by batch. Your decision should follow your claim type and product positioning.
For a deeper explanation, see Full Spectrum Extract vs Standardized Extract.
Does turmeric standardized extract work better with black pepper?
Often, yes for absorption. Piperine has evidence for increasing curcumin bioavailability. That said, higher exposure can also raise interaction risk for some users and medications. Brands should balance efficacy-per-dose with safety messaging and compliance requirements in their target markets.
What’s the biggest QA red flag when buying turmeric extract?
A COA that lists “curcumin” without clarifying total curcuminoids, method, and identity controls is a common red flag. Another major red flag is weak heavy-metal documentation, given known lead-adulteration risks in parts of the supply chain. Treat heavy-metal controls as mandatory.
How to connect turmeric standardized extract benefits to your topical map
If you’re building a standardized-extract cluster, turmeric is an ideal page because it forces you to explain standardization, markers, and QC. Link it to foundational education and comparison pages:
- Standard Herb Extract
- Full Spectrum Extract vs Standardized Extract
- Milk Thistle Standardized Extract vs Whole Herb
- Saw Palmetto Standardized Extract vs Whole Herb
Practical takeaway for B2B buyers
Turmeric standardized extract benefits become real for customers when you control three variables: verified curcuminoid potency, safe and traceable supply, and a formulation that delivers consistent exposure. If you want this article to convert, pair it with a COA example, a spec sheet, and a clear RFQ path.
Trust assets that often improve conversions:
- A sample COA (redacted if needed)
- A product specification sheet
- A third-party testing statement (method + lab name)
Regulatory note: This article discusses general evidence and traditional use. It is not medical advice. Claims and labeling must follow your target market’s rules.

